|
STED enables imaging with sub-diffraction resolution down to below 50 nm. These interactions occur within a few nm, but localizing protein pairs is diffraction-limited to approximately 200 nm. The investigation of minute protein structures and interactions within nanoscopic cellular structures in living cells is challenging but essential for understanding biological processes.įLIM-FRET allows detecting and quantifying protein interactions. Although, these technologies enable to run an instrument of comparable complexity and power without having to spend more time on instrument maintenance than on original scientific content, the MicroTime 200 STED remains an open platform that allows the advanced scientist to easily built upon the open character of the instrument in order to realize highly customized applications.Ĭorrelative FLIM-FRET and STED imaging reveals protein interactions and structure in vivo The instrument gains its exceptional sensitivity and flexibility in combination with unprecedented ease-of-use from a unique fusion of miniaturized and highly sophisticated state-of-the-art technologies. It contains the complete optics and electronics for recording virtually all aspects of the fluorescence dynamics of microscopic samples or femtoliter volumes and with a spatial resolution below 50 nm. The MicroTime 200 STED is a powerful instrument capable of STED microscopy, Fluorecence Corelation Spectroscopy and its daughter techniques as well as Fluorescence Lifetime Imaging (FLIM) with single molecule detection sensitivity. MicroTime 200 STED Time-resolved confocal fluorescence microscope with super-resolution capability PicoQuant offers a complete, turn-key solution for STED super-resolution microscopy Hell talks about the origins of STED microscopy data acquisition unit, e.g., a TCSPC module.phase plate for transforming the STED pulse into a doughnut shape.dichroic mirror (to overlay excitation and depletion lasers and to separate fluorescence signal from excitation light).pulsed laser source for excitation and depletion.In the center of the doughnut, where the STED laser intensity is zero, fluorescence remains unaffected and is detected by single-photon sensitive detectors (e.g., Single Photon Avalanche Diodes, SPADs).Ĭonsequently the essential components of a STED microscope set-up are: By spatially arranging the STED pulse in a doughnut shape using specially designed phase plates, only the fluorescence from molecules at the periphery of the excitation focus is quenched via stimulated emission. The excitation pulse is immediately followed by a depletion pulse, which is red-shifted in frequency to the emission spectrum of the dye. a picosecond pulsed diode laser, is used to excite the fluorescence dye and produces an ordinary diffraction limited focus. The STED method uses pairs of synchronized laser pulses. In that case, collecting data at different observation volume diameters can help to disentangle complex 2D diffusion scenarios in heterogeneous samples such as biological membranes. As STED creates an effectively smaller observation volume, it can also be applied to other methods such as FCS. An even further resolution enhancement is possible by applying time gates to the collected data (gated STED or gSTED). Fluorescence from the remaining excited dye molecules in the center of the excitation focus is then detected and used to form the high resolution images. This intense radiation causes almost all of the excited molecules to return to the ground state. The resolution enhancement is essentially based on switching off the fluorescence of dye molecules by stimulated emission using intense laser light in the outer regions of the diffraction limited excitation focus. 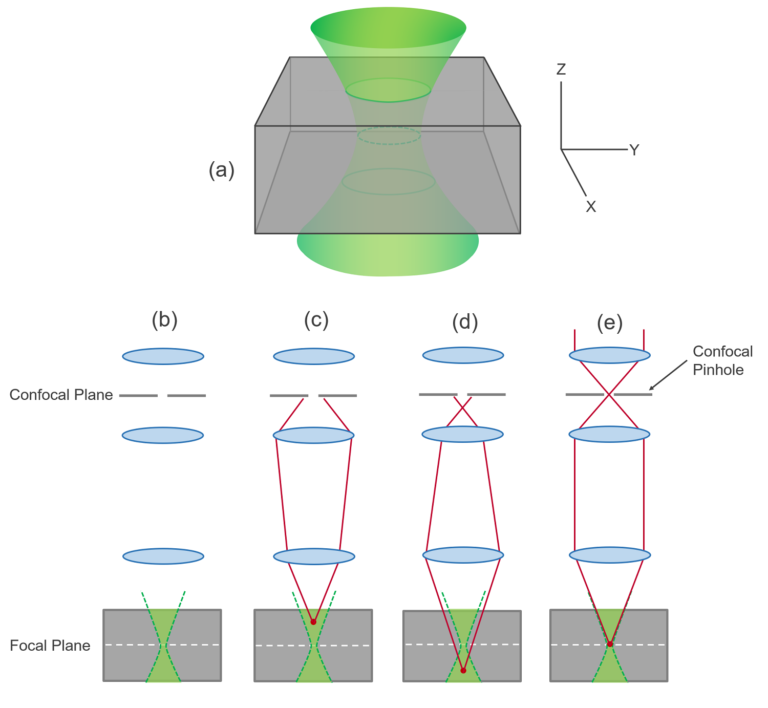
Stimulated emission depletion microscopy (STED) is a fluorescence microscopy technique that overcomes the diffraction limited resolution of confocal microscopes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
